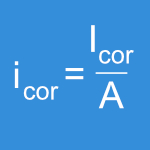PI-60: (ISO 6474; 1994) Implants for surgery – Ceramic materials based on high purity alumina
This wear test is designed as a screening test to evaluate tribologic properties of new designed implant materials or combinations. A ring (20 mm diameter) is oscillated at a 25° rotational movement on a disc (25 mm diameter). Water is used as test fluid but bovine serum or Ringer´s solution can be used optionally. The test is stopped after 100 hours at 1 Hz frequency. The amount of wear is determined by profile measurement of the wear track. EndoLab® GmbH uses a computerized method to gain 3-D results of the wear track. Due to the simplification of the kinematics in-vivo, more complex test procedures like joint simulators have to be used to obtain further information.